What Are PFAS? The Science Behind Forever Chemicals
PFAS have been found in rainwater on every continent, in Arctic ice thousands of miles from any factory, and in the blood of 98% of Americans tested. These synthetic chemicals, invented in the 1940s, have spread so thoroughly through our environment that complete avoidance is now impossible. But what actually are PFAS, and why are they so difficult to get rid of? The answer lies in a single chemical bond—one of the strongest in organic chemistry.
Quick Answer: What Are PFAS?
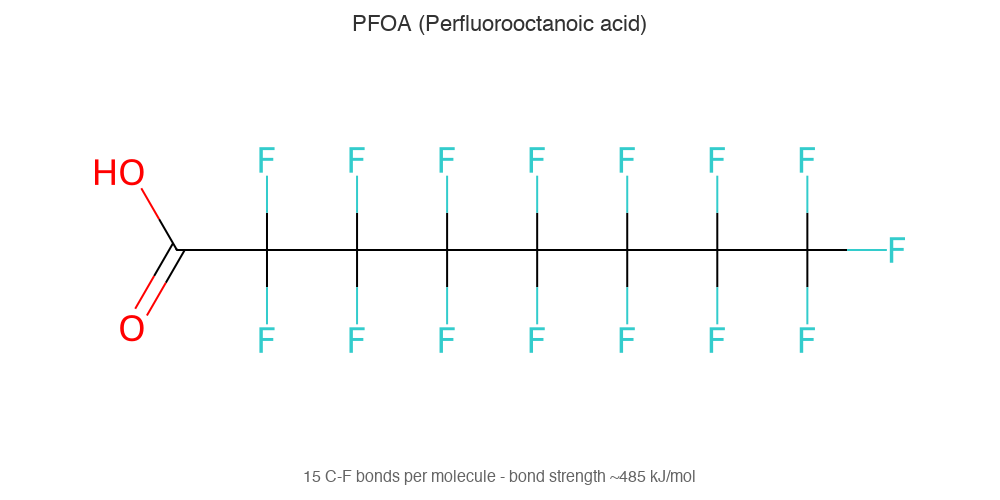
PFAS (per- and polyfluoroalkyl substances) are a group of over 14,000 synthetic chemicals characterised by carbon-fluorine bonds. First developed in the 1940s, they're used in non-stick coatings, waterproof fabrics, food packaging, and firefighting foam. Their exceptional stability—the same property that makes them useful—means they persist in the environment for decades or longer, earning them the name "forever chemicals."
Key Takeaways
- The carbon-fluorine bond (~485 kJ/mol) is one of the strongest in organic chemistry—nothing in nature breaks it down efficiently
- PFAS have been detected in drinking water serving 100+ million Americans, and in rainwater globally
- Health concerns include links to certain cancers, thyroid disease, immune effects, and elevated cholesterol
- Water filters certified to NSF/ANSI Standard 53 or 58 can remove PFAS; boiling water does not
- EPA's 2024 standards set limits of 4 parts per trillion for PFOA and PFOS in drinking water
Why Are PFAS Called Forever Chemicals?
The name isn't hyperbole—it's chemistry. PFAS molecules contain chains of carbon atoms bonded to fluorine atoms, and the carbon-fluorine bond is one of the strongest single bonds in organic chemistry. At approximately 485 kJ/mol, it takes extraordinary energy to break.
To understand why this matters, consider how most pollutants eventually disappear. Organic compounds typically break down through UV light exposure, bacterial metabolism, or chemical reactions with oxygen and water. These processes attack the bonds holding molecules together. But PFAS resist all of them.
Bacteria can't metabolise PFAS because no enzymes have evolved to break carbon-fluorine bonds—these molecules simply don't exist in nature. UV light lacks the energy to cleave C-F bonds efficiently. Chemical oxidation, which degrades most organic pollutants, barely touches them. The same stability that made PFAS revolutionary for non-stick coatings and waterproofing makes them essentially immortal in the environment.
Here's a useful comparison: diesel fuel spilled in soil will largely biodegrade within months to years. PCBs, notorious persistent pollutants banned decades ago, have half-lives measured in years to decades. PFAS? We don't actually know their environmental half-life because we haven't observed significant degradation. Estimates range from decades to centuries—possibly longer.
This creates a ratchet effect. Every kilogram of PFAS manufactured since the 1940s is still out there, somewhere—in groundwater, in ocean sediments, in living organisms. The contamination only accumulates.
Where Did PFAS Come From?
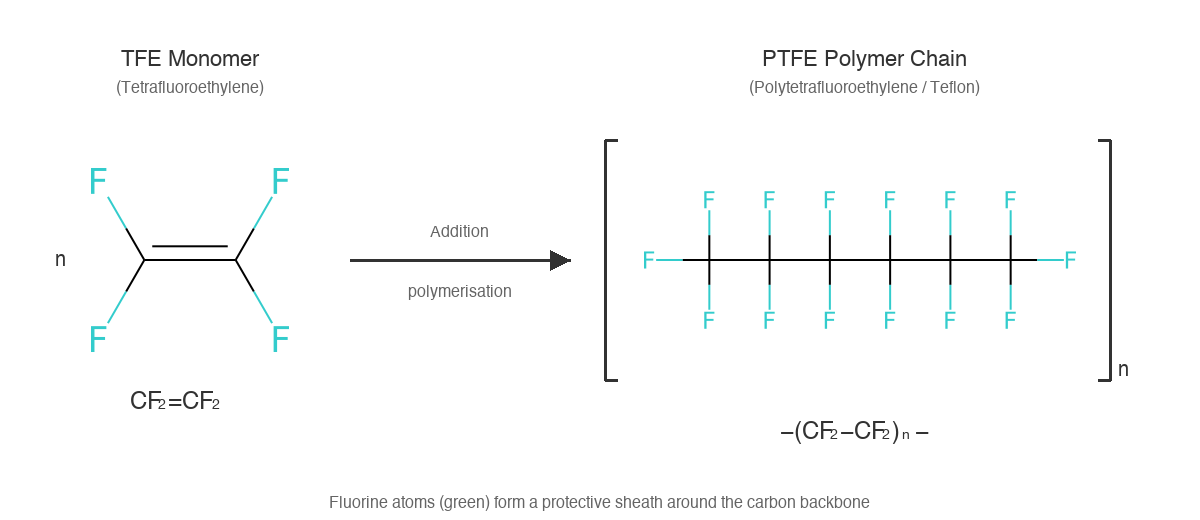
Like many chemical discoveries, PFAS were an accident. In 1938, DuPont chemist Roy Plunkett was attempting to develop new refrigerants when he noticed a waxy white residue in a cylinder of tetrafluoroethylene gas. That residue was polytetrafluoroethylene—PTFE—which DuPont eventually trademarked as Teflon.
PTFE was remarkable: almost nothing stuck to it, it was chemically inert, and it could withstand extreme temperatures. By the 1950s, Teflon-coated cookware was entering American kitchens. Around the same time, 3M developed PFOS (perfluorooctane sulfonate) for their Scotchgard fabric protector, which made carpets and upholstery resistant to stains and water.
Then came the application that would cause the most environmental damage: aqueous film-forming foam (AFFF) for fighting fuel fires. PFAS-based firefighting foams could smother jet fuel and petroleum fires faster than anything else available. The US military adopted them widely, as did civilian airports and oil refineries. Training exercises meant these foams were sprayed repeatedly at the same sites for decades.
By the 1970s, PFAS had become embedded in modern life: cookware, clothing, food packaging, cosmetics, electronics, medical devices. The same properties—water resistance, oil resistance, heat resistance, chemical stability—made them useful for almost everything. And for decades, almost no one asked what happened when these chemicals reached the environment.
How Teflon Works: The Chemistry of PTFE
Understanding why Teflon is non-stick reveals why PFAS are forever chemicals—it's the same chemistry.
PTFE starts as tetrafluoroethylene (TFE), a simple monomer: two carbon atoms connected by a double bond, with four fluorine atoms attached. When polymerised, the double bond opens and TFE molecules link together in an addition polymerisation reaction:
n CF₂=CF₂ → −(CF₂−CF₂)n−
Addition polymerisation: monomers join without losing atoms
The result is a long chain of repeating −CF₂−CF₂− units—potentially thousands of them. Here's what makes this structure special: the fluorine atoms completely surround the carbon backbone like a protective sheath. Fluorine is the most electronegative element, but paradoxically, C-F bonds have very low polarisability. This means the surface presents almost no opportunity for other molecules to interact with it. Water can't hydrogen-bond to it. Oils can't form van der Waals interactions with it. Food literally has nothing to grab onto—hence, non-stick.
The same property that prevents eggs from sticking to your pan prevents bacteria from metabolising PFAS in the environment. The fluorine sheath that repels water and oil also repels the enzymes that would normally break down organic molecules. The C-F bond's strength—the source of PTFE's heat resistance and chemical inertness—is precisely why these compounds persist for decades. Non-stick and forever are two descriptions of the same molecular reality.
Where Are PFAS Found Today?
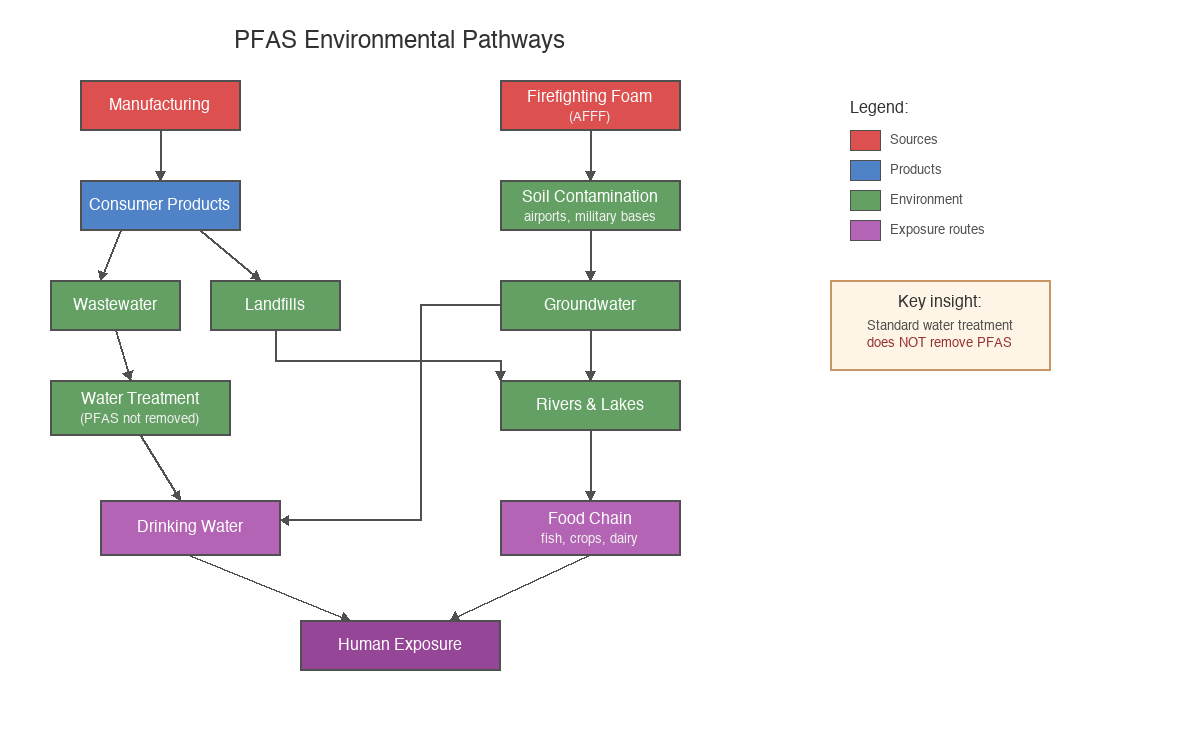
The short answer: everywhere. The long answer is more unsettling.
Drinking water is the primary exposure route for most people. PFAS contamination has been detected in water supplies serving over 100 million Americans, with the highest concentrations near manufacturing facilities, military bases, and airports. Groundwater contamination is particularly problematic because PFAS are mobile in soil—they migrate with water flow and can travel miles from the original source.
Food packaging represents a direct route into our bodies. Grease-resistant wrappers, microwave popcorn bags, fast-food containers, and pizza boxes often contain PFAS to prevent oil from soaking through. The chemicals migrate into food, particularly hot or greasy food.
Cookware and kitchen products include non-stick pans, baking sheets, and cooking utensils coated with PTFE or similar fluoropolymers. While intact coatings are generally considered stable, overheating or scratching can release PFAS or PFAS-containing particles.
Clothing and outdoor gear marketed as waterproof or water-resistant often relies on PFAS treatments. Gore-Tex, until recently, used PFAS-based membranes. Rain jackets, hiking boots, tents, and ski gear frequently contain these compounds.
Cosmetics and personal care products including foundations, mascaras, lipsticks, and sunscreens may contain PFAS for smooth application or water resistance. Look for ingredients containing "fluoro" or "PTFE" on labels.
The broader environment now contains PFAS essentially everywhere. Studies have detected these chemicals in rainwater globally at levels exceeding safety guidelines, in Arctic wildlife far from any industrial source, in agricultural soils irrigated with contaminated water, and in the food chain from fish to dairy to vegetables. The contamination is no longer localised—it's planetary.
Are PFAS Dangerous? What the Research Shows
The honest answer is: we're still learning, but what we know is concerning.
The most-studied PFAS compounds—PFOA and PFOS—have been linked to several health effects in human epidemiological studies:
- Cancer: Increased risk of kidney cancer and testicular cancer in highly exposed populations, such as workers at manufacturing plants and communities with contaminated water
- Thyroid disease: Associations with thyroid hormone disruption and thyroid disease
- Immune system effects: Reduced vaccine antibody response, particularly in children—one of the most consistent findings across studies
- Reproductive effects: Links to pregnancy-induced hypertension, reduced birth weight, and developmental effects
- Elevated cholesterol: One of the most robust associations, seen even at relatively low exposure levels
- Liver effects: Elevated liver enzymes indicating liver stress
Important caveats apply. Most research has focused on PFOA and PFOS, but there are thousands of PFAS compounds with varying structures and potentially different toxicity profiles. Dose matters—workers with high occupational exposure show clearer health effects than the general population with lower environmental exposure. And epidemiological studies show associations, not necessarily causation.
What makes PFAS particularly concerning is bioaccumulation. Unlike many chemicals that are metabolised and excreted relatively quickly, PFAS build up in the body over time. The half-life of PFOA in human blood is approximately 3-4 years; for PFOS, it's 4-5 years. This means ongoing exposure leads to steadily increasing body burden.
The regulatory response has been to phase out PFOA and PFOS in favour of shorter-chain PFAS and alternative compounds. However, these replacements are less studied, and emerging research suggests some may share similar properties. We may be repeating the cycle of introducing chemicals faster than we can understand their effects—a pattern seen with synthetic compounds throughout industrial chemistry.
How Do Scientists Detect PFAS in Water?
This is where I can offer some first-hand perspective. During my chemistry degree, I developed HPLC-MS methods for quantifying PFAS in environmental water samples as part of my dissertation research. The analytical chemistry involved is both impressive and sobering.
HPLC-MS (high-performance liquid chromatography coupled with mass spectrometry) is the standard method for PFAS analysis. Here's how it works in accessible terms:
First, water samples undergo solid-phase extraction (SPE). Because PFAS concentrations are often in parts per trillion, you need to concentrate them. A large volume of water is passed through a cartridge containing specialised materials that capture PFAS while letting most other compounds through. The captured PFAS are then washed off with a small volume of solvent, effectively concentrating them by a factor of 100-1000x.
The concentrated extract goes into the HPLC system. Under high pressure, the sample is pushed through a narrow column packed with particles. Different PFAS compounds interact differently with the column material, causing them to travel at different speeds. This separates the mixture—PFOS might exit after 8 minutes, PFOA after 10 minutes, and so on.
As each compound exits the column, it enters the mass spectrometer. Here, molecules are ionised (given an electrical charge) and then sorted by their mass-to-charge ratio. Each PFAS compound produces a characteristic fragmentation pattern—a molecular fingerprint that allows definitive identification. By comparing the signal intensity to known standards, we can quantify exactly how much of each PFAS is present.
Modern methods can detect PFAS at concentrations below 1 part per trillion. To put that in perspective: one part per trillion is equivalent to one second in 32,000 years, or one drop of water in 20 Olympic swimming pools. This sensitivity is both reassuring—we can find contamination—and alarming—we keep finding it.
The analytical approach shares principles with methods used in other fields. Supercritical CO₂ extraction, used in coffee decaffeination, exploits similar selective solubility principles, though for very different purposes. Understanding these separation techniques is fundamental to both removing unwanted compounds and detecting harmful ones.
Can PFAS Be Removed from Water?
Yes, but with significant caveats. Several treatment technologies can remove PFAS from drinking water:
Activated carbon adsorbs PFAS onto its highly porous surface. Granular activated carbon (GAC) filters are effective, particularly for longer-chain PFAS like PFOA and PFOS. The carbon eventually becomes saturated and must be replaced or regenerated—at which point you have concentrated PFAS waste that still needs disposal.
Ion exchange resins are specialised materials designed to swap ions—in this case, capturing negatively charged PFAS molecules. These can be more effective than activated carbon for some PFAS, particularly shorter-chain variants that GAC struggles with. Again, the resins eventually saturate and require regeneration or replacement.
Reverse osmosis and nanofiltration physically separate PFAS by pushing water through membranes with pores small enough to block these molecules. These are highly effective but energy-intensive and produce a concentrated waste stream.
The fundamental problem with all these methods is that they don't destroy PFAS—they just move them from water into a concentrated form that still needs managing. You've protected the drinking water, but created hazardous waste.
Destruction technologies that actually break the C-F bond are emerging but not yet widely deployed:
- High-temperature incineration (above 1000°C) can mineralise PFAS, but requires specialised facilities and careful emission controls
- Supercritical water oxidation uses water at extreme temperature and pressure to break down PFAS
- Electrochemical oxidation and various advanced oxidation processes show promise in research settings
- Sonochemical destruction uses ultrasound to break bonds
These destruction methods are expensive, energy-intensive, and not yet available at scale for most contaminated water supplies. For now, we're largely stuck with removal and containment rather than true elimination.
How Can I Reduce My PFAS Exposure?
Complete avoidance is impossible—PFAS are too widespread. But reducing exposure is worthwhile and practical:
Water: Check if your water utility tests for PFAS (many now do) and review the results. If levels are concerning or unknown, consider a water filter certified to NSF/ANSI Standard 53 or 58 for PFAS reduction. Reverse osmosis systems under the sink are highly effective. Quality activated carbon filters help but vary in effectiveness. Note that standard Brita-style pitcher filters may reduce some PFAS but aren't typically certified for comprehensive removal.
Cookware: If using non-stick pans, keep heat at medium or below—most concerns arise from overheating. Never preheat empty non-stick pans. Replace any pans with scratched, flaking, or damaged coatings. Alternatives include cast iron, stainless steel, or ceramic-coated cookware.
Food packaging: Limit use of grease-resistant fast food wrappers, microwave popcorn bags, and takeaway containers, especially for hot or greasy foods. Transfer food to glass or ceramic containers for storage and reheating.
Personal care products: Check cosmetics, particularly long-wear makeup and waterproof products, for ingredients containing "fluoro," "PTFE," or "perfluoro." Several databases now track PFAS in cosmetics.
Clothing and gear: When buying waterproof clothing, look for PFAS-free alternatives—many outdoor brands are transitioning away from fluorinated treatments. Be more cautious with older "waterproof" items, which likely contain PFAS.
Be realistic: You cannot eliminate all PFAS exposure, and attempting to do so will drive you to distraction. Focus on the biggest sources—drinking water, heavily used cookware, daily-use cosmetics—where changes make the most difference.
What's Being Done About PFAS?
Regulatory action is accelerating, though critics argue it's decades overdue:
United States: In April 2024, the EPA established the first legally enforceable national drinking water standards for PFAS, setting limits of 4 parts per trillion for PFOA and PFOS individually, with additional limits for other PFAS compounds. The EPA has also designated PFOA and PFOS as hazardous substances under CERCLA (Superfund law), enabling cleanup enforcement and potentially holding manufacturers liable for contamination.
European Union: Moving toward a broad restriction on all PFAS for non-essential uses—potentially the most comprehensive regulatory action globally. Several EU countries have already implemented strict limits.
State-level action: Several US states have enacted PFAS restrictions stricter than federal standards, including bans on PFAS in food packaging, cosmetics, and firefighting foam.
Industry response: Major manufacturers have phased out PFOA and PFOS, but replacements remain contentious. Some replacement compounds, like GenX, have raised their own health concerns. The chemical industry argues that PFAS are essential for many applications and that blanket restrictions could have unintended consequences for critical industries including semiconductors and medical devices.
Litigation: PFAS manufacturers face thousands of lawsuits from water utilities, states, and individuals. 3M agreed to a settlement of up to $12.5 billion with public water systems in 2023. DuPont/Chemours has faced similar actions. These settlements may fund cleanup efforts but don't solve the underlying contamination.
The Bottom Line
PFAS represent a sobering case study in the unintended consequences of industrial chemistry. The same carbon-fluorine bond that made these compounds so useful—stable, unreactive, water-resistant, heat-resistant—makes them essentially permanent once released into the environment.
Understanding the chemistry helps make sense of the headlines. PFAS aren't called forever chemicals because of marketing or alarmism—they're called forever chemicals because the C-F bond genuinely resists every natural degradation process we know of. The contamination we've created over 80 years will outlast us all.
That said, understanding the problem is the first step toward managing it. We now have analytical methods sensitive enough to detect PFAS at vanishingly small concentrations. We have treatment technologies that can remove them from drinking water. We have regulatory frameworks taking shape. And we have growing awareness driving both consumer choices and industry reformulation.
The challenge ahead is enormous: cleaning up existing contamination, finding alternatives for essential applications, preventing new releases, and ultimately developing technologies that can actually destroy these persistent molecules. It's a problem we created, and it's one we'll be managing for generations.
Further Reading & Viewing
For a deeper dive into the corporate history and current contamination crisis, Veritasium's documentary How One Company Secretly Poisoned the Planet is highly recommended. The film Dark Waters (2019) dramatises attorney Rob Bilott's legal battle against DuPont, revealing how long the company knew about PFAS toxicity before it became public.
For technical detail on the regulatory standards, the Federal Register's final PFAS rule provides the complete text of the 2024 EPA drinking water regulation.
Frequently Asked Questions
Is Teflon still made with PFAS?
PFOA, the PFAS used in Teflon manufacturing, was phased out by 2015. However, Teflon itself (PTFE) is a fluoropolymer—a long-chain molecule containing carbon-fluorine bonds. While PTFE is considered stable when intact, concerns have shifted to replacement chemicals used in production and potential releases when cookware is overheated above 260°C (500°F).
Should I throw away my non-stick pans?
Not necessarily. Undamaged non-stick cookware used at appropriate temperatures (medium heat, never preheated empty) is generally considered safe. Replace pans with scratched, flaking, or damaged coatings. If you want to avoid fluoropolymers entirely, switch to cast iron, stainless steel, or ceramic alternatives.
Does boiling water remove PFAS?
No. Boiling water does not remove PFAS—it actually concentrates them as water evaporates. PFAS are thermally stable and won't break down at cooking temperatures. Effective removal requires activated carbon filtration, reverse osmosis, ion exchange, or nanofiltration systems.
Are PFAS in all drinking water?
PFAS have been detected in water supplies serving over 100 million Americans, but not all water is contaminated. Levels are highest near industrial facilities, military bases, airports, and firefighting training sites. Check your water utility's testing results or consider independent laboratory testing if concerned.
What level of PFAS is safe?
The EPA's 2024 standards set enforceable limits of 4 parts per trillion for PFOA and PFOS. Whether any level is truly "safe" remains debated—PFAS bioaccumulate over time, so even low ongoing exposure builds body burden. The precautionary approach is minimising exposure where practical while avoiding unnecessary alarm.
Do water filters remove PFAS?
Some do. Look for filters certified to NSF/ANSI Standard 53 (activated carbon) or Standard 58 (reverse osmosis) specifically for PFAS reduction. Reverse osmosis is most effective. Quality carbon block filters help but vary. Standard pitcher filters may reduce some PFAS but typically lack certification for comprehensive removal.
Written by Miles Christou, chemistry graduate who developed HPLC-MS methods for PFAS quantification in environmental water samples