How Do Soaps Work? The Chemistry of Surfactants and Cleaning
You wash your hands dozens of times a day, but have you ever stopped to wonder how soap actually works? It seems almost magical: you rub a slippery bar on your hands, add water, and suddenly grease, dirt, and bacteria wash away. The chemistry behind this everyday miracle is both elegant and surprisingly complex. At its heart is a simple principle—soap molecules have a split personality, loving both water and oil simultaneously—but the implications of this molecular schizophrenia revolutionised human hygiene.
Quick Answer: How Does Soap Work?
Soap works because its molecules are amphiphilic—they have a hydrophobic (water-repelling) tail that dissolves in oils and fats, and a hydrophilic (water-loving) head that dissolves in water. When you wash, soap molecules surround oil and dirt particles with their tails buried in the grease and their heads facing outward into the water. This allows oily dirt to be suspended in water and rinsed away. Above a certain concentration, soap molecules form structures called micelles that trap oils inside, making cleaning even more effective.
Key Takeaways
- Soap molecules are surfactants—they have a hydrophobic hydrocarbon tail (~12-18 carbons) and a hydrophilic carboxylate head (COO⁻)
- Traditional soap is made through saponification: fats/oils + strong base (NaOH or KOH) → soap + glycerol
- Micelles form above the critical micelle concentration (CMC), creating structures that trap oils and enable effective cleaning
- Modern detergents use synthetic surfactants (anionic, cationic, nonionic, amphoteric) designed for specific applications and hard water compatibility
- Soap works primarily through mechanical removal, not by killing microbes—proper handwashing technique matters more than antibacterial additives
What Is Soap? The Chemistry of Saponification
Before understanding how soap cleans, we need to understand what soap actually is—and that requires looking at its chemical structure.
Traditional soap is the sodium or potassium salt of a long-chain fatty acid. It's made through a chemical reaction called saponification—a process humans have used for thousands of years, though they didn't understand the chemistry until the 19th century.
Saponification starts with fats or oils (triglycerides)—molecules consisting of three fatty acid chains attached to a glycerol backbone. When you treat these fats with a strong base like sodium hydroxide (NaOH, also known as lye or caustic soda), the ester bonds holding the fatty acids to glycerol break apart in a hydrolysis reaction:
Fat/Oil + [NaOH] → Soap + Glycerol
Triglyceride + 3 [NaOH] → 3 Fatty Acid Salts + Glycerol
The result is three molecules of soap (fatty acid salts) and one molecule of glycerol. This is an example of base-catalysed ester hydrolysis—the hydroxide ion attacks the carbonyl carbon of the ester bond, breaking it and releasing the fatty acid, which immediately reacts with sodium ions to form the salt.
The structure of a typical soap molecule looks like this: a long hydrocarbon chain (typically 12-18 carbon atoms) with a carboxylate group (−COO⁻ Na⁺) at one end. For example, sodium stearate (made from stearic acid, an 18-carbon saturated fatty acid) is a common soap component:
[CH₃(CH₂)₁₆COO⁻ Na⁺]
Sodium stearate - 18 carbon atoms in the tail, carboxylate head
This structure—a long, nonpolar hydrocarbon tail attached to a polar, ionic head—is what makes soap work. The tail is hydrophobic (water-fearing) because it's made of carbon and hydrogen, which don't form hydrogen bonds with water. The head is hydrophilic (water-loving) because it's a charged carboxylate group that interacts strongly with water molecules.
This dual nature makes soap an amphiphilic molecule—from Greek amphi (both) and philos (loving). It simultaneously loves water and oil, existing comfortably at the interface between them. This is the fundamental property that enables cleaning.
How Soap Cleans: The Mechanics of Dirt Removal
Now to the practical question: how does this molecular structure translate into cleaning?
Most dirt isn't water-soluble. Grease, oils, wax, many organic compounds—these stick to your skin or clothing because they're hydrophobic. Water alone can't wash them away because there's no chemical affinity between water (polar) and oils (nonpolar). This is why rinsing your greasy hands with just water doesn't work—the oil and water don't mix.
Soap solves this problem by acting as a molecular bridge between the oil and the water.
When you apply soap to your oily hands, the hydrophobic tails of soap molecules dissolve into the oil or grease. At the same time, the hydrophilic heads remain dissolved in the water. The soap molecules orient themselves at the oil-water interface with their tails buried in the oil and their heads sticking out into the water.
As you rub your hands together (mechanical action is crucial—soap alone sitting on your skin doesn't clean much), the oil breaks up into smaller and smaller droplets. Each droplet becomes surrounded by soap molecules, creating a structure where the oil is encased in a shell of soap with all the hydrophilic heads pointing outward into the water.
This creates an emulsion—a suspension of tiny oil droplets dispersed in water. The oil droplets are now effectively "disguised" as water-soluble particles because they're covered in the hydrophilic heads of soap molecules. When you rinse, these soap-coated oil droplets wash away with the water rather than re-depositing on your skin.
This process is called emulsification, and it's the primary mechanism of how soap cleans. But there's more to the story—the formation of micelles takes cleaning to another level.
Micelle Formation: Why Concentration Matters
At low concentrations, soap molecules exist as individual ions dissolved in water. But something remarkable happens when you increase the soap concentration beyond a certain threshold called the critical micelle concentration (CMC).
Above the CMC, soap molecules spontaneously organise themselves into structures called micelles. A micelle is a spherical cluster of typically 50-100 surfactant molecules arranged with their hydrophobic tails pointing inward toward the centre and their hydrophilic heads pointing outward toward the water.
This arrangement makes thermodynamic sense. In water, the hydrophobic tails are uncomfortable—they disrupt the hydrogen bonding network of water molecules, which is energetically unfavourable (this is the hydrophobic effect). By clustering together with tails on the inside, the surfactant molecules minimise the contact between their hydrophobic portions and water, while keeping their hydrophilic heads happily solvated in the aqueous environment.
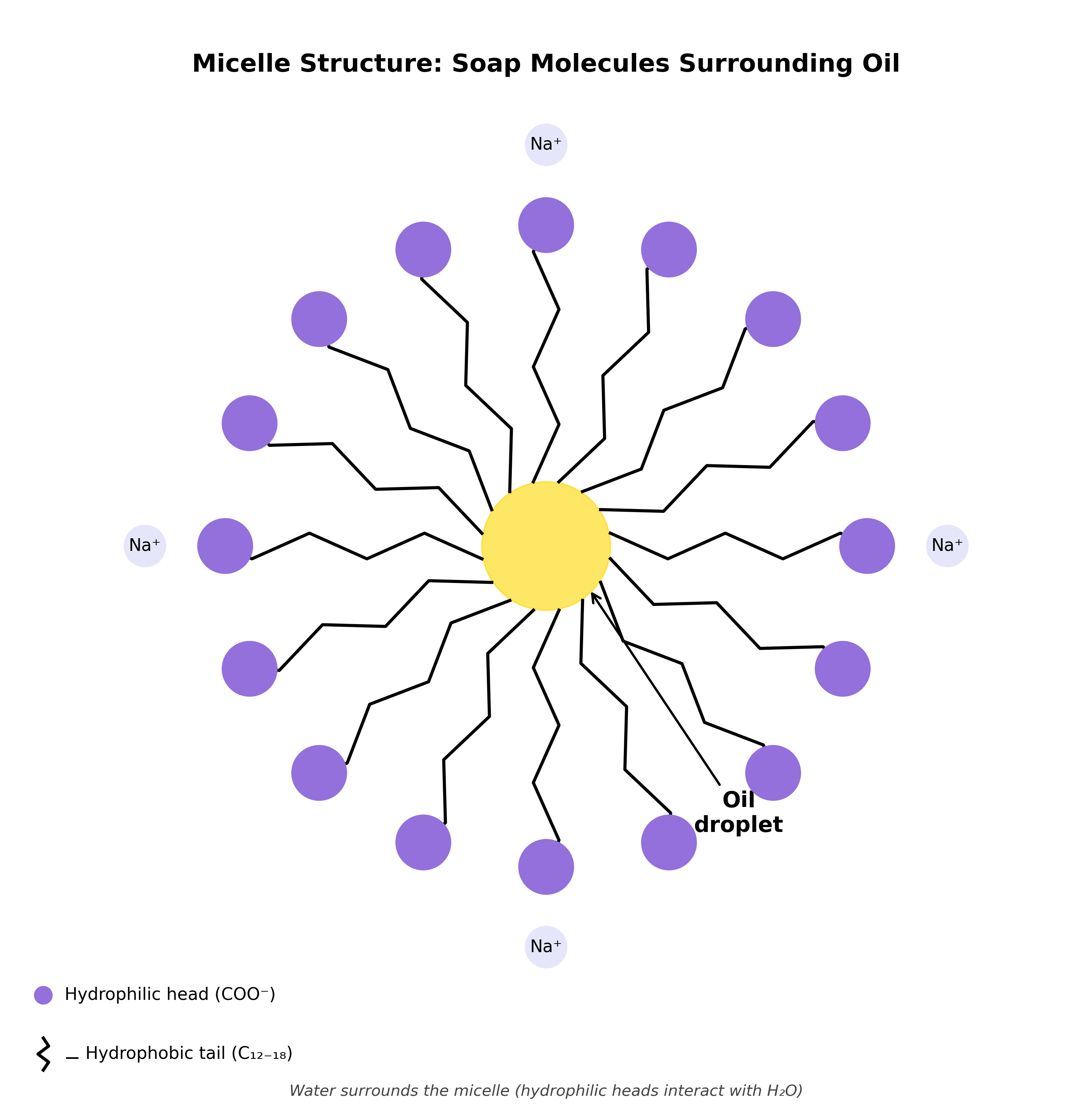
The interior of a micelle is a hydrophobic environment—essentially a tiny droplet of oil-like material suspended in water. This is where the real cleaning power comes in.
When oil or grease is present, it dissolves into the hydrophobic core of the micelles. The micelle structure essentially acts as a molecular cage, trapping oils and nonpolar dirt inside while remaining soluble in water because of the hydrophilic shell. This is called solubilisation—making something water-insoluble (oil) effectively water-soluble by hiding it inside a micelle.
This is why soap becomes dramatically more effective above the CMC. Below the CMC, you're relying primarily on emulsification—soap molecules coating oil droplets. Above the CMC, you have thousands of micelles actively solubilising oils into their cores, plus emulsification still occurring. The cleaning action becomes much more efficient.
The CMC varies depending on the specific surfactant, temperature, and presence of other substances (salts, other surfactants, etc.), but for typical soaps it's in the range of 1-10 millimoles per litre. This is why concentrated soap solutions clean better than very dilute ones—you need to exceed the CMC to get micelle formation.
Surfactants: Beyond Traditional Soap
Soap is just one type of surfactant—a contraction of "surface-active agent." Surfactants are amphiphilic molecules that adsorb at interfaces (like the air-water or oil-water boundary) and alter the properties of those interfaces, particularly surface tension.
Traditional soap has limitations, particularly in hard water. Hard water contains dissolved calcium (Ca²⁺) and magnesium (Mg²⁺) ions, which react with soap's carboxylate groups to form insoluble precipitates—the familiar grey scum that forms in hard water:
2 [C₁₇H₃₅COO⁻ Na⁺] + Ca²⁺ → [C₁₇H₃₅COO⁻]₂Ca²⁺ + 2 Na⁺
Soluble sodium soap + calcium ion → insoluble calcium soap (scum)
This wastes soap and prevents effective cleaning. To solve this, chemists developed synthetic detergents—surfactants designed to avoid this problem while retaining cleaning ability.
Surfactants are classified based on the charge of their hydrophilic head group:
1. Anionic Surfactants
These have a negatively charged head group. Traditional soap (carboxylate, −COO⁻) is anionic, but modern anionic surfactants often use sulfonate (−SO₃⁻) or sulfate (−OSO₃⁻) head groups instead:
- Linear alkylbenzene sulfonates (LAS): The most common surfactants in laundry detergents. These have a benzene ring with a sulfonate group and a linear alkyl chain (typically C₁₀-C₁₄). Unlike carboxylate soaps, sulfonate groups don't precipitate with calcium and magnesium, making LAS effective in hard water.
- Sodium lauryl sulfate (SLS): Also called sodium dodecyl sulfate, this is ubiquitous in personal care products—shampoos, toothpastes, body washes. It's an effective cleanser and foaming agent, though it can be irritating to sensitive skin at high concentrations.
- Sodium laureth sulfate (SLES): An ethoxylated version of SLS, slightly milder and also extremely common in consumer products.
Anionic surfactants are excellent at removing oily dirt and are generally the workhorses of cleaning formulations, making up the bulk of most laundry and dishwashing detergents.
2. Cationic Surfactants
These have a positively charged head group, typically a quaternary ammonium compound (four organic groups bonded to nitrogen, giving it a permanent positive charge). The most common structure is R−N⁺(CH₃)₃ Cl⁻, where R is a long alkyl chain.
Cationic surfactants don't clean as well as anionic ones, but they have other useful properties. They're often used as:
- Fabric softeners: The positive charge causes them to adsorb onto negatively charged fabric surfaces, creating a soft, lubricating layer
- Hair conditioners: Hair is slightly negatively charged (especially when damaged), so cationic surfactants bind to it, smoothing the cuticle and reducing static
- Disinfectants: Many quaternary ammonium compounds ("quats") have antimicrobial properties because they disrupt bacterial cell membranes
Cationic and anionic surfactants don't mix well—they can neutralise each other and precipitate out. This is why you don't wash clothes with fabric softener in the same cycle as detergent, or why you shouldn't mix different cleaning products without understanding their chemistry.
3. Nonionic Surfactants
These have no charge on the head group—instead, they rely on polar functional groups like alcohols or ethers for their hydrophilic character. Common examples include:
- Alcohol ethoxylates: Long-chain alcohols with multiple ethylene oxide units (−O−CH₂−CH₂−) attached. The ether oxygens are polar and hydrogen-bond with water, providing hydrophilicity without charge.
- Alkyl polyglucosides (APGs): Made from sugars (glucose) and fatty alcohols. These are biodegradable, mild, and increasingly popular in "eco-friendly" formulations.
Nonionic surfactants have several advantages: they work well in both hard and soft water (no charge to react with metal ions), they're generally milder and less irritating than ionic surfactants, and they can be mixed with other surfactant types without issues. They're often used in combination formulations to boost performance.
4. Amphoteric (Zwitterionic) Surfactants
These contain both positive and negative charges in the same molecule. The most common are betaines (like cocamidopropyl betaine, found in many shampoos) and amphoacetates.
Amphoteric surfactants are exceptionally mild and are often used in baby shampoos and products for sensitive skin. They're compatible with all other surfactant types and can act as foam boosters in formulations.
Why Does Soap Produce Foam?
Foam is perhaps the most visible sign that soap is "working," but it's largely decorative—foam doesn't actually do the cleaning. Understanding why it forms, though, reveals more about surfactant chemistry.
Foam is a dispersion of gas (air) in a liquid (water). Normally, water doesn't foam much because it has high surface tension. Surface tension arises because water molecules at the surface are attracted to molecules below them but not above (where there's only air), creating an inward pull that minimises surface area.
When you add surfactant, the molecules adsorb at the air-water interface with their hydrophobic tails pointing toward the air and hydrophilic heads toward the water. This arrangement is energetically favourable—the tails are escaping the water they don't like, and the heads remain solvated.
By occupying the interface, surfactants dramatically reduce surface tension. Lower surface tension means it's easier to create new surface area—which is exactly what happens when you form bubbles. The surfactant layer also stabilises the bubbles by forming a flexible film that resists rupture.
However, foam and cleaning ability aren't correlated. Some excellent detergents produce little foam (automatic dishwasher detergent is deliberately low-foaming to prevent overflow), while some poor cleaners foam extensively. Consumer perception has driven manufacturers to add foam boosters to products like shampoo because people expect lots of foam, even though it's unnecessary for cleaning.
The exception is in applications where mechanical action is limited—like handwashing dishes or personal hygiene. Foam can help by holding surfactant at the surface being cleaned and providing a visual indicator of where soap has been applied. But fundamentally, it's the surfactant molecules doing the work, not the bubbles.
Do Soaps Kill Germs?
This is a common misconception: soap doesn't kill most bacteria or viruses—it removes them mechanically.
The same amphiphilic properties that allow soap to dissolve oils also allow it to disrupt lipid membranes. Bacteria are surrounded by cell membranes made of phospholipids—molecules that are themselves amphiphilic, with hydrophobic tails and hydrophilic heads arranged in a bilayer. Enveloped viruses (like influenza and SARS-CoV-2) have similar lipid membranes.
When exposed to sufficient concentrations of surfactants, these membranes can be disrupted or dissolved, effectively inactivating the microbes. However, at the concentrations used in normal handwashing, the primary mechanism is physical removal—the soap helps lift bacteria and viruses from skin surfaces, and then they're washed away with water. The mechanical action of rubbing your hands together is crucial for this.
This is why proper handwashing technique matters more than the type of soap. The CDC recommends washing with soap and water for at least 20 seconds, scrubbing all surfaces of the hands—palm, back, between fingers, under nails. The duration and thoroughness physically dislodge and remove microbes.
"Antibacterial" soaps containing additives like triclosan were marketed as superior for years, but research showed they offered no significant advantage over regular soap for routine handwashing. In 2016, the FDA banned triclosan and 18 other antibacterial agents from consumer soaps, citing insufficient evidence of benefit and concerns about antimicrobial resistance.
In healthcare settings, antiseptic hand rubs containing 60-95% alcohol are different—these do kill microbes through protein denaturation and membrane disruption, and are effective when soap and water aren't available. But for everyday use, regular soap with proper technique is highly effective at reducing microbial load through removal.
Modern Detergent Formulations: It's Not Just Surfactants
If you look at the ingredients list on a modern laundry detergent, you'll see it's far more complex than just surfactants. Commercial formulations typically include:
- Builders: Compounds that improve cleaning by softening water (binding calcium and magnesium ions that would otherwise interfere with surfactants). Sodium carbonate (washing soda) and sodium citrate are common builders. Phosphates were formerly used but are now largely banned due to environmental concerns (they cause eutrophication in waterways).
- Enzymes: Biological catalysts that break down specific stains. Proteases digest protein stains (blood, grass, food), amylases break down starches, lipases target fats and oils, and cellulases remove loosened fibres to prevent pilling and maintain brightness. Similar enzymatic approaches are used in other applications, from brewing to decaffeination.
- Bleaching agents: Oxidisers that remove coloured stains by breaking the chromophores (colour-causing structures) in molecules. Sodium percarbonate releases hydrogen peroxide when dissolved, which oxidises stains. Optical brighteners (fluorescent whitening agents) don't actually clean—they absorb UV light and emit blue light, making fabrics appear whiter by counteracting yellowing.
- Anti-redeposition agents: These prevent dirt that's been removed from fabric from settling back onto it during the wash. Carboxymethyl cellulose is common—it binds to dirt particles and keeps them suspended in the wash water.
- Fragrance and dyes: Purely aesthetic. Many people are sensitive to these, hence the growing market for "free and clear" formulations.
Industrial and institutional cleaning products take formulation even further, with specialised surfactant blends, pH buffers, corrosion inhibitors, and other additives designed for specific applications—degreasing, floor care, sanitising, and so on.
The Environmental Angle: Biodegradability and Aquatic Toxicity
All these surfactants and cleaning agents eventually go down the drain and into wastewater treatment systems. This raises important questions about environmental impact.
Early synthetic detergents in the 1940s-50s used branched alkyl chains that bacteria couldn't easily degrade, leading to persistent foaming in rivers and wastewater treatment plants. The industry eventually switched to linear alkyl chains, which are readily biodegradable.
Modern surfactants are generally designed for biodegradability, but this varies. Linear alkylbenzene sulfonates (LAS) degrade within days to weeks in aerobic conditions. Alcohol ethoxylates also biodegrade readily. Quaternary ammonium compounds are more persistent and can be toxic to aquatic organisms at low concentrations.
Regulations like the EU Detergents Regulation mandate that surfactants in consumer products meet biodegradability criteria. However, "biodegradable" doesn't mean "harmless"—surfactants can still be toxic to aquatic life during the time they persist, and metabolites (breakdown products) can themselves have environmental effects.
This parallels concerns with other persistent chemicals. Just as PFAS contamination demonstrates the risks of using chemicals faster than we understand their environmental fate, surfactant formulation involves balancing performance with environmental responsibility—a challenge that continues to drive innovation in green chemistry.
The Bottom Line
Soap is deceptively simple—just molecules with one part that likes water and another that likes oil. But this simple structural feature has profound implications: it enables emulsification of oils, formation of micelles that solubilise dirt, reduction of surface tension, and mechanical removal of microbes.
Understanding how soap works reveals why certain practices matter: why you need mechanical action (rubbing, agitation) in addition to the chemical action of surfactants; why soap concentration matters (you need to exceed the CMC for maximum effectiveness); why hard water interferes with traditional soap but not synthetic detergents; and why "antibacterial" claims on consumer soaps are largely marketing rather than meaningful benefit.
The evolution from ancient soap (animal fat + wood ash) to modern multi-component detergent formulations reflects the intersection of chemistry, engineering, and consumer demand. Whether you're hand-washing dishes, laundering clothes, or scrubbing your hands, you're harnessing the chemistry of amphiphilic molecules—a chemical principle that's been changing the world one clean surface at a time for thousands of years.
Frequently Asked Questions
What is the difference between soap and detergent?
Soaps are made from natural fats and oils through saponification with alkali (NaOH or KOH), producing fatty acid salts. Detergents are synthetic surfactants made from petroleum or plant-derived feedstocks through chemical processes. The key practical difference: soaps form insoluble scum with the calcium and magnesium in hard water, while most detergents use sulfonate or sulfate head groups that don't react with these ions, making them effective in hard water.
How does soap remove oil and grease?
Soap molecules are amphiphilic—they have a hydrophobic tail (typically 12-18 carbons) that dissolves in oils and a hydrophilic head (carboxylate group for traditional soap) that dissolves in water. The tails embed in grease while heads remain in water, allowing oil to emulsify into tiny droplets that rinse away. Above the critical micelle concentration, micelles form with oil-friendly cores that trap grease inside while remaining water-soluble on the outside.
What are micelles and why do they matter?
Micelles are spherical aggregates of surfactant molecules (typically 50-100 molecules) that form above a threshold concentration (the critical micelle concentration, or CMC). They arrange with hydrophobic tails pointing inward and hydrophilic heads outward, creating an oil-friendly core surrounded by a water-friendly shell. This structure solubilises oils by trapping them inside, dramatically increasing cleaning effectiveness compared to simple emulsification. Micelle formation is what makes soap a powerful cleaner, not just a surface-active agent.
Why does soap produce foam?
Surfactants reduce surface tension by adsorbing at the air-water interface with hydrophobic tails toward air and hydrophilic heads toward water. This makes it energetically easier to create new surface area (bubbles) and stabilises them against rupture. However, foam doesn't clean—it's a side effect of surfactant chemistry. Some highly effective cleaners foam minimally (dishwasher detergent), while some poor cleaners foam extensively. Foam's main benefit is visual feedback and, in some applications, holding surfactant in place during cleaning.
What makes hard water incompatible with soap?
Hard water contains dissolved Ca²⁺ and Mg²⁺ ions. These react with soap's carboxylate groups (−COO⁻) to form insoluble calcium and magnesium salts—the sticky grey scum you see in bathtubs and sinks. This wastes soap and prevents effective cleaning. Synthetic detergents solve this by using sulfonate (−SO₃⁻) or sulfate (−OSO₃⁻) head groups that don't precipitate with these ions, or by including builders (like sodium citrate) that bind calcium and magnesium ions before they can react with surfactants.
Are antibacterial soaps more effective than regular soap?
For everyday use, no. Regular soap works primarily through mechanical removal—surfactants lift bacteria and viruses from skin, and they're washed away with water. The physical action of proper handwashing (20+ seconds, scrubbing all surfaces) matters more than antibacterial additives. The FDA banned triclosan and 18 other antibacterial agents from consumer soaps in 2016 due to insufficient evidence of benefit over regular soap and concerns about antimicrobial resistance. In healthcare settings, specific antiseptic formulations (like alcohol-based hand rubs) serve important purposes, but for home use, regular soap with proper technique is equally effective.
Written by Miles Christou, chemistry graduate